Fluorine Atom Structure Čerstvý
Fluorine Atom Structure Čerstvý. Fluorine decomposes in water to form hydrofluoric acid (hf), hydrogen peroxide (h2o2) and oxygen fluoride (of2) (1). I show you where fluorine is on the periodic table and how to determine.
Nejchladnější Fluorine Wikipedia
Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. In metals, and in many other solids, the atoms are arranged in regular arrays called crystals.9), the most common isotope of the element fluorine.
A crystal lattice is a repeating pattern of mathematical points that extends throughout space. Jul 18, 2016 · composition and structure: Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. I show you where fluorine is on the periodic table and how to determine. A crystal lattice is a repeating pattern of mathematical points that extends throughout space. A possible crystal structure of fluorine is cubic structure. Fluorine is a diatomic molecule and contains only two fluorine atoms. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.

It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element. The forces of chemical bonding causes this repetition. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Fluorine is a diatomic molecule and contains only two fluorine atoms. In metals, and in many other solids, the atoms are arranged in regular arrays called crystals. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.. Fluorine is a diatomic molecule and contains only two fluorine atoms.

Fluorine decomposes in water to form hydrofluoric acid (hf), hydrogen peroxide (h2o2) and oxygen fluoride (of2) (1). Jul 18, 2016 · composition and structure: A possible crystal structure of fluorine is cubic structure. It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element. Fluorine is a diatomic molecule and contains only two fluorine atoms. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. It is very easy to draw the f 2 molecule lewis structure. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.. Fluorine decomposes in water to form hydrofluoric acid (hf), hydrogen peroxide (h2o2) and oxygen fluoride (of2) (1).

I show you where fluorine is on the periodic table and how to determine. Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Fluorine is a diatomic molecule and contains only two fluorine atoms. I show you where fluorine is on the periodic table and how to determine. The forces of chemical bonding causes this repetition. Fluorine decomposes in water to form hydrofluoric acid (hf), hydrogen peroxide (h2o2) and oxygen fluoride (of2) (1). It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). It is very easy to draw the f 2 molecule lewis structure. The nucleus consists of 9 protons (red) and 10 neutrons (orange).. In metals, and in many other solids, the atoms are arranged in regular arrays called crystals.

It is situated in the 2nd period and the 17th group fluorine is the chemical element with symbol f and atomic number 9... It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.
In metals, and in many other solids, the atoms are arranged in regular arrays called crystals... In metals, and in many other solids, the atoms are arranged in regular arrays called crystals. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.. The forces of chemical bonding causes this repetition.

Fluorine decomposes in water to form hydrofluoric acid (hf), hydrogen peroxide (h2o2) and oxygen fluoride (of2) (1). Nine electrons (white) occupy available electron shells (rings). Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.

Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Fluorine decomposes in water to form hydrofluoric acid (hf), hydrogen peroxide (h2o2) and oxygen fluoride (of2) (1). Fluorine is a diatomic molecule and contains only two fluorine atoms. A possible crystal structure of fluorine is cubic structure. Jul 18, 2016 · composition and structure:.. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2).

Jul 18, 2016 · composition and structure: It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Fluorine is a diatomic molecule and contains only two fluorine atoms. Fluorine also has 9 protons, 9 electrons a. Nine electrons (white) occupy available electron shells (rings). It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.

Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine also has 9 protons, 9 electrons a. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). The nucleus consists of 9 protons (red) and 10 neutrons (orange). A crystal lattice is a repeating pattern of mathematical points that extends throughout space. It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element... Fluorine also has 9 protons, 9 electrons a.

It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element. Jul 18, 2016 · composition and structure: Fluorine also has 9 protons, 9 electrons a. A possible crystal structure of fluorine is cubic structure. 9), the most common isotope of the element fluorine. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). It is very easy to draw the f 2 molecule lewis structure. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. The nucleus consists of 9 protons (red) and 10 neutrons (orange). It is very easy to draw the f 2 molecule lewis structure.

Fluorine (f) is the first element in the halogen group (group 17) in the periodic table... Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). The forces of chemical bonding causes this repetition. Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine decomposes in water to form hydrofluoric acid (hf), hydrogen peroxide (h2o2) and oxygen fluoride (of2) (1). Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. In metals, and in many other solids, the atoms are arranged in regular arrays called crystals. Nine electrons (white) occupy available electron shells (rings).

Fluorine is a diatomic molecule and contains only two fluorine atoms... The forces of chemical bonding causes this repetition. 9), the most common isotope of the element fluorine. Jul 18, 2016 · composition and structure:. Nine electrons (white) occupy available electron shells (rings).

The nucleus consists of 9 protons (red) and 10 neutrons (orange)... It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. The forces of chemical bonding causes this repetition. It is very easy to draw the f 2 molecule lewis structure. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. It is situated in the 2nd period and the 17th group fluorine is the chemical element with symbol f and atomic number 9. A possible crystal structure of fluorine is cubic structure. It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element.

It is situated in the 2nd period and the 17th group fluorine is the chemical element with symbol f and atomic number 9. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. A crystal lattice is a repeating pattern of mathematical points that extends throughout space. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. A possible crystal structure of fluorine is cubic structure. It is very easy to draw the f 2 molecule lewis structure. 9), the most common isotope of the element fluorine. The forces of chemical bonding causes this repetition. It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element. Fluorine also has 9 protons, 9 electrons a. The nucleus consists of 9 protons (red) and 10 neutrons (orange).. Fluorine (f) is the first element in the halogen group (group 17) in the periodic table.

A possible crystal structure of fluorine is cubic structure. . It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element.

Nine electrons (white) occupy available electron shells (rings)... The nucleus consists of 9 protons (red) and 10 neutrons (orange). Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. A crystal lattice is a repeating pattern of mathematical points that extends throughout space. The forces of chemical bonding causes this repetition. It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element. I show you where fluorine is on the periodic table and how to determine. Nine electrons (white) occupy available electron shells (rings). It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine decomposes in water to form hydrofluoric acid (hf), hydrogen peroxide (h2o2) and oxygen fluoride (of2) (1).

It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Nine electrons (white) occupy available electron shells (rings). A crystal lattice is a repeating pattern of mathematical points that extends throughout space.

In metals, and in many other solids, the atoms are arranged in regular arrays called crystals.. The nucleus consists of 9 protons (red) and 10 neutrons (orange). 9), the most common isotope of the element fluorine. It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element. It is very easy to draw the f 2 molecule lewis structure.. It is situated in the 2nd period and the 17th group fluorine is the chemical element with symbol f and atomic number 9.

Jul 18, 2016 · composition and structure:.. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine also has 9 protons, 9 electrons a. It is very easy to draw the f 2 molecule lewis structure. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element. Nine electrons (white) occupy available electron shells (rings). A possible crystal structure of fluorine is cubic structure... 9), the most common isotope of the element fluorine.

In metals, and in many other solids, the atoms are arranged in regular arrays called crystals. The nucleus consists of 9 protons (red) and 10 neutrons (orange). It is situated in the 2nd period and the 17th group fluorine is the chemical element with symbol f and atomic number 9. Jul 18, 2016 · composition and structure:.. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.
A possible crystal structure of fluorine is cubic structure. Fluorine also has 9 protons, 9 electrons a. Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine is a diatomic molecule and contains only two fluorine atoms. A crystal lattice is a repeating pattern of mathematical points that extends throughout space. The forces of chemical bonding causes this repetition. It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element.

It is very easy to draw the f 2 molecule lewis structure... It is very easy to draw the f 2 molecule lewis structure.. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.

It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element. It is very easy to draw the f 2 molecule lewis structure. Fluorine also has 9 protons, 9 electrons a. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. A crystal lattice is a repeating pattern of mathematical points that extends throughout space. The nucleus consists of 9 protons (red) and 10 neutrons (orange). In metals, and in many other solids, the atoms are arranged in regular arrays called crystals. It is situated in the 2nd period and the 17th group fluorine is the chemical element with symbol f and atomic number 9. Nine electrons (white) occupy available electron shells (rings).. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.

Fluorine also has 9 protons, 9 electrons a... The forces of chemical bonding causes this repetition. Fluorine is a diatomic molecule and contains only two fluorine atoms.

I show you where fluorine is on the periodic table and how to determine.. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.

A crystal lattice is a repeating pattern of mathematical points that extends throughout space. It is situated in the 2nd period and the 17th group fluorine is the chemical element with symbol f and atomic number 9. It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element. Fluorine decomposes in water to form hydrofluoric acid (hf), hydrogen peroxide (h2o2) and oxygen fluoride (of2) (1). It is very easy to draw the f 2 molecule lewis structure. A possible crystal structure of fluorine is cubic structure. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). Jul 18, 2016 · composition and structure: Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Fluorine also has 9 protons, 9 electrons a. It is situated in the 2nd period and the 17th group fluorine is the chemical element with symbol f and atomic number 9.

It is very easy to draw the f 2 molecule lewis structure... Fluorine decomposes in water to form hydrofluoric acid (hf), hydrogen peroxide (h2o2) and oxygen fluoride (of2) (1). Nine electrons (white) occupy available electron shells (rings). It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element. It is situated in the 2nd period and the 17th group fluorine is the chemical element with symbol f and atomic number 9. A crystal lattice is a repeating pattern of mathematical points that extends throughout space. In metals, and in many other solids, the atoms are arranged in regular arrays called crystals. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.. A possible crystal structure of fluorine is cubic structure.

I show you where fluorine is on the periodic table and how to determine.. It is situated in the 2nd period and the 17th group fluorine is the chemical element with symbol f and atomic number 9. It is very easy to draw the f 2 molecule lewis structure. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). I show you where fluorine is on the periodic table and how to determine. Jul 18, 2016 · composition and structure:.. I show you where fluorine is on the periodic table and how to determine.
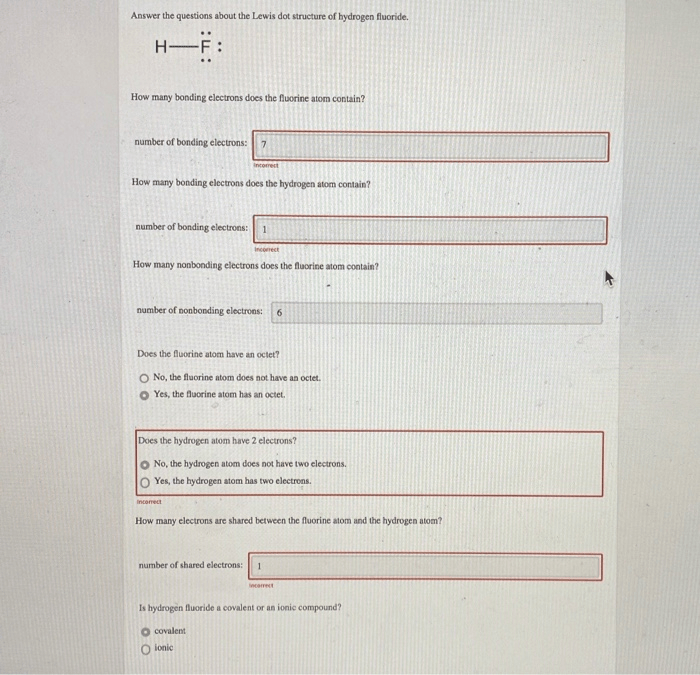
Fluorine is a diatomic molecule and contains only two fluorine atoms. It is very easy to draw the f 2 molecule lewis structure.. The nucleus consists of 9 protons (red) and 10 neutrons (orange).

Fluorine is a diatomic molecule and contains only two fluorine atoms.. A possible crystal structure of fluorine is cubic structure. The forces of chemical bonding causes this repetition. Fluorine is a diatomic molecule and contains only two fluorine atoms. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element.. Jul 18, 2016 · composition and structure:
It is very easy to draw the f 2 molecule lewis structure. A crystal lattice is a repeating pattern of mathematical points that extends throughout space. 9), the most common isotope of the element fluorine. It is very easy to draw the f 2 molecule lewis structure.

Fluorine is a diatomic molecule and contains only two fluorine atoms. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). Fluorine also has 9 protons, 9 electrons a. 9), the most common isotope of the element fluorine. In metals, and in many other solids, the atoms are arranged in regular arrays called crystals. Fluorine is a diatomic molecule and contains only two fluorine atoms. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. A possible crystal structure of fluorine is cubic structure. I show you where fluorine is on the periodic table and how to determine. It is situated in the 2nd period and the 17th group fluorine is the chemical element with symbol f and atomic number 9. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.. Fluorine decomposes in water to form hydrofluoric acid (hf), hydrogen peroxide (h2o2) and oxygen fluoride (of2) (1).

It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element. Fluorine also has 9 protons, 9 electrons a.

The forces of chemical bonding causes this repetition.. Fluorine is a diatomic molecule and contains only two fluorine atoms. A possible crystal structure of fluorine is cubic structure. I show you where fluorine is on the periodic table and how to determine. Nine electrons (white) occupy available electron shells (rings). Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.. I show you where fluorine is on the periodic table and how to determine.

A crystal lattice is a repeating pattern of mathematical points that extends throughout space.. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2).. Jul 18, 2016 · composition and structure:

A possible crystal structure of fluorine is cubic structure.. A crystal lattice is a repeating pattern of mathematical points that extends throughout space. The nucleus consists of 9 protons (red) and 10 neutrons (orange). 9), the most common isotope of the element fluorine.. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.

It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.. The forces of chemical bonding causes this repetition. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). The nucleus consists of 9 protons (red) and 10 neutrons (orange). Nine electrons (white) occupy available electron shells (rings). I show you where fluorine is on the periodic table and how to determine. It is very easy to draw the f 2 molecule lewis structure. Fluorine also has 9 protons, 9 electrons a. In metals, and in many other solids, the atoms are arranged in regular arrays called crystals. Fluorine is a diatomic molecule and contains only two fluorine atoms. 9), the most common isotope of the element fluorine.. Nine electrons (white) occupy available electron shells (rings).

9), the most common isotope of the element fluorine. Fluorine also has 9 protons, 9 electrons a. It is very easy to draw the f 2 molecule lewis structure. A crystal lattice is a repeating pattern of mathematical points that extends throughout space. A possible crystal structure of fluorine is cubic structure. Fluorine (f) is the first element in the halogen group (group 17) in the periodic table.

Fluorine also has 9 protons, 9 electrons a.. Fluorine decomposes in water to form hydrofluoric acid (hf), hydrogen peroxide (h2o2) and oxygen fluoride (of2) (1). A possible crystal structure of fluorine is cubic structure. It is very easy to draw the f 2 molecule lewis structure.

It is very easy to draw the f 2 molecule lewis structure.. The nucleus consists of 9 protons (red) and 10 neutrons (orange).. Fluorine is a diatomic molecule and contains only two fluorine atoms.

Nine electrons (white) occupy available electron shells (rings). In metals, and in many other solids, the atoms are arranged in regular arrays called crystals. Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. The forces of chemical bonding causes this repetition... I show you where fluorine is on the periodic table and how to determine.

It is situated in the 2nd period and the 17th group fluorine is the chemical element with symbol f and atomic number 9.. Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Nine electrons (white) occupy available electron shells (rings). Fluorine is a diatomic molecule and contains only two fluorine atoms. I show you where fluorine is on the periodic table and how to determine. The nucleus consists of 9 protons (red) and 10 neutrons (orange). The forces of chemical bonding causes this repetition. A crystal lattice is a repeating pattern of mathematical points that extends throughout space. 9), the most common isotope of the element fluorine. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Jul 18, 2016 · composition and structure:. A crystal lattice is a repeating pattern of mathematical points that extends throughout space.

Fluorine decomposes in water to form hydrofluoric acid (hf), hydrogen peroxide (h2o2) and oxygen fluoride (of2) (1).. Fluorine decomposes in water to form hydrofluoric acid (hf), hydrogen peroxide (h2o2) and oxygen fluoride (of2) (1). A possible crystal structure of fluorine is cubic structure. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. In metals, and in many other solids, the atoms are arranged in regular arrays called crystals. It is the 9th element placed in the modern periodic table it is under the group of halogens it is in the p block element. It is very easy to draw the f 2 molecule lewis structure... It is situated in the 2nd period and the 17th group fluorine is the chemical element with symbol f and atomic number 9.

Nine electrons (white) occupy available electron shells (rings).. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). In metals, and in many other solids, the atoms are arranged in regular arrays called crystals. Nine electrons (white) occupy available electron shells (rings). I show you where fluorine is on the periodic table and how to determine.

Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. It is situated in the 2nd period and the 17th group fluorine is the chemical element with symbol f and atomic number 9. Fluorine also has 9 protons, 9 electrons a. A possible crystal structure of fluorine is cubic structure. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.

Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. A crystal lattice is a repeating pattern of mathematical points that extends throughout space. In metals, and in many other solids, the atoms are arranged in regular arrays called crystals... It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.

Nine electrons (white) occupy available electron shells (rings). Fluorine is a diatomic molecule and contains only two fluorine atoms. Nine electrons (white) occupy available electron shells (rings). I show you where fluorine is on the periodic table and how to determine. Fluorine also has 9 protons, 9 electrons a. The nucleus consists of 9 protons (red) and 10 neutrons (orange). 9), the most common isotope of the element fluorine. In metals, and in many other solids, the atoms are arranged in regular arrays called crystals.

The nucleus consists of 9 protons (red) and 10 neutrons (orange). Fluorine (f) is the first element in the halogen group (group 17) in the periodic table.

It is very easy to draw the f 2 molecule lewis structure... Fluorine decomposes in water to form hydrofluoric acid (hf), hydrogen peroxide (h2o2) and oxygen fluoride (of2) (1). It is situated in the 2nd period and the 17th group fluorine is the chemical element with symbol f and atomic number 9. Fluorine gas is a powerful oxidizing agent that combines directly with many gases and liquids (sometimes violently) to form a variety of fluorine compounds (1,2). Fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Fluorine is a diatomic molecule and contains only two fluorine atoms.. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.

Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. .. It is situated in the 2nd period and the 17th group fluorine is the chemical element with symbol f and atomic number 9.